Lunit gets Canadian licenses for AI radiology solutions
Mobi Health News
JUNE 21, 2022
This follows last year's FDA approvals in the US.
This site uses cookies to improve your experience. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country, we will assume you are from the United States. Select your Cookie Settings or view our Privacy Policy and Terms of Use.
Cookies and similar technologies are used on this website for proper function of the website, for tracking performance analytics and for marketing purposes. We and some of our third-party providers may use cookie data for various purposes. Please review the cookie settings below and choose your preference.
Used for the proper function of the website
Used for monitoring website traffic and interactions
Cookies and similar technologies are used on this website for proper function of the website, for tracking performance analytics and for marketing purposes. We and some of our third-party providers may use cookie data for various purposes. Please review the cookie settings below and choose your preference.

Mobi Health News
JUNE 21, 2022
This follows last year's FDA approvals in the US.

Bill of Health
NOVEMBER 29, 2023
By Vincent Joralemon Spravato, the first FDA-approved psychedelic therapy, just outsold Viagra. Pfizer’s successful clinical trials for ED followed, leading to FDA approval of sildenafil citrate, marketed as “Viagra,” in 1998. This became Spravato, the first FDA-approved psychedelic therapy. Initially, the public is hesitant.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Mobi Health News
FEBRUARY 13, 2025
The bill would allow AI to be classified as a 'practitioner licensed by law' to administer FDA-approved drugs.

Healthcare It News
MAY 12, 2023
" Since launching its claims platform, a series of new capabilities, including prior authorizations and denials management, has been added. miRNA-based cancer diagnostics Ovarian cancer is highly lethal in part because of how difficult it has historically been to diagnose.

Healthcare It News
APRIL 27, 2023
" According to an email from a Philips representative, the announcement marks the first commercialized use of the technology following two years of field testing, publication of a peer-reviewed clinical study and the FDA’s classification of RATE as a wellness device. The score has proven to be indicative of onset of infections.

HIT Consultant
NOVEMBER 24, 2022
First, let’s examine the new regulatory framework the FDA established. – Prescription hearing aids will require a consumer to have a prescription for a hearing aid from a physician, audiologist or a licensed hearing instrument specialist. These OTC hearing aids do not require a prescription. . yet is largely untreated.

HIT Consultant
MAY 5, 2025
– The model uniquely pairs licensed physical therapists with Laina, the company’s proprietary WebAI assistant. This plan is then delivered and monitored through Laina, an FDA-registered, HIPAA-compliant WebAI assistant. Patients access Laina conveniently on their own devices without needing additional hardware or downloads.

Bill of Health
OCTOBER 31, 2022
For instance, a study conducted in 2005 by the American Association for Advancement of Sciences revealed that 40% of respondents reported problems securing licenses for carrying out research; 58% reported delays due to licensing requirements; and 28% reported abandoning their research due to licensing issues.

Healthcare IT Today
MARCH 26, 2025
Federal and state agencies, including the Drug Enforcement Administration ( DEA ) and the Food and Drug Administration (FDA), are shaping the future of telehealth through evolving policies. The FDA also plays a key role in overseeing online pharmacies to ensure they comply with federal drug distribution laws.

Bill of Health
APRIL 4, 2024
In a similar vein, FDA only considers medical AI that falls in one of its existing regulatory categories (most often medical device), and even then by way of Congressional action and FDA’s own interpretation of its authority, and its discretion only regulates a subset of medical AI. A different example has to do with how the U.S.

Healthcare IT Today
MARCH 31, 2025
.” Epicore’s suite of sweat-sensing wearable solutions includes the Gx Sweat Patch , commercialized in partnership with PepsiCo and Gatorade, the Discovery Patch Sweat Collection System (FDA Class I device), and the Connected Hydration wearable and cloud platform tailored for industrial workers. Follow Epicore on LinkedIn.

Verisys
APRIL 8, 2025
However, most clients opt for the FACIS Level 3 background check as it is all-inclusive and protects them from employing individuals or vendors who are sanctioned by a state-level licensing board. Treasury Dept U.S. State Dept Which Level of FACIS Search Does Your Organization Need? The post What is FACIS?
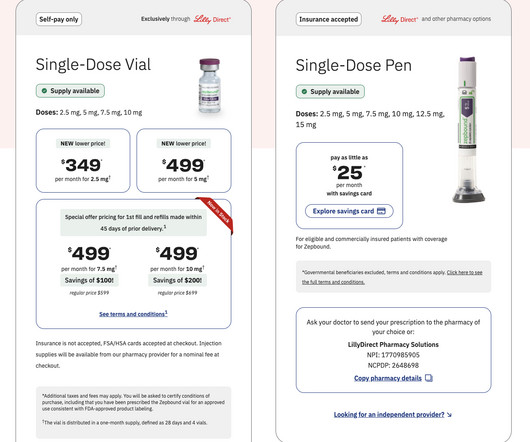
HIT Consultant
MARCH 6, 2025
– The collaboration will enable eligible Teladoc Health members enrolled in the Comprehensive Weight Care Program to receive convenient and affordable access to this FDA-approved medication. Personalized support: Tailored guidance and support to help members achieve their weight loss goals.

Bill of Health
JUNE 5, 2023
medical licensing exam , diagnose illnesses , and even outshine human doctors on measures of perceived empathy , raising many questions about how AI will reshape health care as we know it. Food and Drug Administration (FDA). In the field of medicine, ChatGPT already has been reported to ace the U.S.

Hall Render
MAY 11, 2022
Earlier this year, the Food and Drug Administration (“FDA”) announced a Proposed Rule regarding national standards for the licensing of prescription drug wholesale distributors (“Wholesale Distributors”) and third-party logistics providers (“3PLs”). Accordingly, FDA is proposing the withdrawal and replacement of the current 21 C.F.R.

Healthcare It News
DECEMBER 1, 2021
As more and more wearable devices are becoming common in the technology, they have even started to get some FDA approvals as well. But one priority to address is the licensing-based requirement. But in addition to that, we do see a lot of trends toward increased use of remote patient monitoring.
Healthcare IT Today
JANUARY 15, 2025
The trend is already clear, with FDA approvals for AI-related products rising significantly, from 650 devices in 2023 to 950 in 2024. The Consolidation Appropriations Act of 2023 requires strict cybersecurity measures for medical devices authorized by the FDA.

C&M Health Law
FEBRUARY 1, 2022
Food & Drug Administration (FDA) published a notice in the Federal Register announcing the availability of a final guidance for industry and FDA staff entitled “Principles of Premarket Pathways for Combination Products”. The final guidance is available on the FDA’s website. On January 31, 2022, the U.S.

Healthcare IT News - Telehealth
AUGUST 6, 2024
UpScriptHealth also works with other medical device companies to increase access – for example, it powers telehealth consultations for Spark Biomedical's Sparrow Ascent, the only FDA-cleared, drug-free, non-addictive, non-invasive, wearable treatment technology for the relief of opioid withdrawal symptoms.

HIT Consultant
DECEMBER 5, 2024
The platform includes licensed physical therapists, physicians, and board-certified health coaches to help individuals treat and prevent joint and muscle pain. Access to the FDA-cleared Enso wearable device for non-invasive, non-addictive pain relief, tailored for those experiencing significant movement-related pain.

Health Law Advisor
SEPTEMBER 12, 2023
The HHS recommendation is predicated, via the FDA, on a scientific and medical evaluation of marijuana, using a statutorily required eight-factor analysis. In addition, only certain components of FDA’s eight-factor analysis bind the DEA. This effectively allows the DEA to adopt a different outcome than the FDA.

Healthcare It News
NOVEMBER 12, 2021
CommonSpirit is developing the digital capabilities and clinical and administrative processes to provide these services, which will enable patients to have greater control over their care, empower education, increase engagement, target improved clinical outcomes and further support population health management. Twitter: @SiwickiHealthIT.

Hall Render
OCTOBER 18, 2024
Food and Drug Administration (“FDA”) issued two compliance policies that outlined its strategy for enforcing the drug distribution security requirements established by the Drug Supply Chain Security Act (“DSCSA”), which were set to take effect on November 27, 2023.

HIT Consultant
DECEMBER 15, 2022
Founded in 2021 with collaboration with Mayo Clinic, Anumana has licensed Mayo Clinic AI-ECG algorithms for low ejection fraction, pulmonary hypertension, and hyperkalemia, all of which have received Breakthrough Device Designation from the U.S. Anumana Background.

HIT Consultant
JUNE 10, 2024
What You Should Know: – ALZpath , a leader in Alzheimer’s disease diagnostics, announced a licensing agreement with Roche for their innovative pTau217 antibody. ALZpath’s licensing strategy, making their pTau217 antibody available across various settings, aims to democratize access to this vital technology.

Healthcare IT Today
JANUARY 24, 2024
They are still waiting on FDA clearance (like most in this space), but the concept is so simple and beautiful. Of course, these companies can’t say this is what they’re able to do until they get FDA clearance, but the early results seem promising. Here’s a look at how their demo worked for me.

Hall Render
JULY 29, 2022
Food and Drug Administration (“FDA”) issued guidance (“Guidance”) regarding the “Importation of Prescription Drugs” final rule (the “Final Rule”), which became effective November 30, 2020. SIPs are reviewed and authorized by the FDA and managed by States or Indian Tribes (“SIP Sponsors”). In May 2022, the U.S. Brief Discussion.

HIT Consultant
NOVEMBER 23, 2021
XRHealth has virtual clinics throughout the United States that treat patients remotely with VR/AR-based therapy while they are examined by a licensed XRHealth therapist. XRHealth Background.

Healthcare IT Today
JANUARY 19, 2024
About XRHealth XRHealth operates state-of-the-art therapeutic care Virtual Rooms, utilizing proprietary FDA and CE-registered medical Extended Reality (XR) technology (virtual and augmented reality).

HIT Consultant
DECEMBER 14, 2021
In order to prescribe buprenorphine, clinicians need a separate license from the federal government called an x-waiver, which only 5 percent have. At its core is buprenorphine (Suboxone), a medication used to treat withdrawal and cravings while also mitigating overdose risk. A major barrier to expanding access to MAT in the U.S.

Hall Render
SEPTEMBER 15, 2023
In June 2023, the Food and Drug Administration (“FDA”) released Draft Guidance outlining its current view of the psychedelic research landscape. While the FDA does not define psychedelics, it indicated that the shorthand term “psychedelic” includes “classic psychedelics,” which are typically understood to be 5-HT2 agonists (e.g.,

HIT Consultant
SEPTEMBER 21, 2022
The company operates state-of-the-art therapeutic care Virtual Clinics, utilizing proprietary FDA and CE registered medical Extended Reality (XR) technology (virtual and augmented reality). XRHealth is revolutionizing healthcare, bringing patient care into the Metaverse.

Mobi Health News
MARCH 1, 2018
Qualcomm Life announced an exclusive licensing agreement with AlertWatch, creator of an FDA-cleared intelligence care software that assists in the care of patients in the operating room. The deal will give Qualcomm Life the exclusive rights to sell AlertWatch, which the company plans to showcase at HIMSS 2018.

Healthcare IT News - Telehealth
FEBRUARY 5, 2024
Patients with mental health issues frequently must wait many months before getting seen by a licensed clinician who can prescribe psychotropic medication. You contend telepsychiatry will continue to grow because it enables access to mental healthcare – which is a big challenge today.

HIT Consultant
JULY 22, 2022
The Oncousticssolutions for ultrasound will be submitted for regulatory approval in the United States (FDA 510(k)), Canada (Health Canada medical device license) and the European Union (CE Mark). The OnX Liver Assessment Solution has not been cleared for clinical use and is For Investigational Use Only.

Verisys
JULY 8, 2024
Stabilization Period: The FDA has established a one-year stabilization period from November 2023 to November 2024 to allow trading partners to fully implement and troubleshoot their electronic systems without disrupting the supply chain. Without centralized licensing and DSCSA standards, bad actors can easily put patient safety at risk.

Healthcare IT Today
OCTOBER 7, 2024
SBOMs are a kind of inventory list that contains top-level components, sub-components, and dependencies, both direct and transitive, along with the associated licenses and security vulnerabilities. By delivering an SBOM to the medical organizations that use the devices, device manufacturers can assure users of the safety of the devices.
Healthcare IT News - Telehealth
MAY 6, 2020
It also equips providers with Eko’s FDA-cleared AI to assist in remote screening for AFib, heart murmurs, tachycardia and bradycardia. In addition, the company is awaiting FDA approval for an onboard ECG feature for spot detection of atrial fibrillation. iRhythm Technologies.

Healthcare Law Blog
MAY 31, 2022
Medical devices are regulated by the Food and Drug Administration (“FDA”) and many of the applications and technologies used to deliver or access healthcare in the metaverse may be classified as a medical device. [3] How healthcare providers are licensed in the metaverse is also an open question. Medical Device Regulations. 1, 2021).

Verisys
JULY 8, 2024
Stabilization Period: The FDA has established a one-year stabilization period from November 2023 to November 2024 to allow trading partners to fully implement and troubleshoot their electronic systems without disrupting the supply chain. Without centralized licensing and DSCSA standards, bad actors can easily put patient safety at risk.

HIT Consultant
DECEMBER 15, 2021
Aidoc has seven FDA-cleared solutions and has won numerous awards including the prestigious UCSF ‘Best New Health Application of AI’ award and TIME’s 50 most genius companies. BELKIN Laser , a clinical-stage medical device company developing a disruptive automated one-second laser treatment for glaucoma, raised $13.75

Dot Compliance
JANUARY 22, 2025
ISO 9001, FDA 21 CFR Part 11, ISO 13485 ). Cost and ROI Initial Investment: Consider the upfront costs of the eQMS, including licensing fees, implementation costs, and hardware requirements. Customization Options: Look for an eQMS that offers customization capabilities to tailor the system to your specific needs. GDPR, CCPA).

Bill of Health
APRIL 7, 2023
Research centers and companies that received government funds are typically allowed to manage their contractual and licensing terms independently with their commercial partners. Despite this, the study notes that public research grants do not usually include safeguards to enhance public access or affordability.

Healthcare IT Today
OCTOBER 18, 2023
A licensed clinician reviews patient responses and provides a diagnosis, care plan, prescriptions, or connects the patient to a different modality or setting for care when clinically appropriate. MDLIVE health coaching is powered by Welldoc ® , a digital health leader in chronic care with FDA-cleared solutions.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content